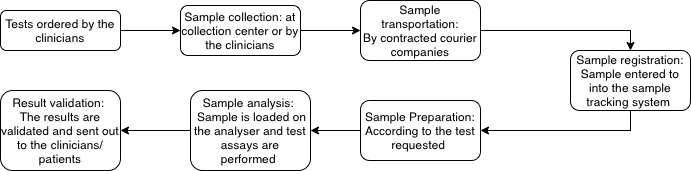
Behind every diagnosis, treatment decision and clinical outcome lies an invisible but critical system: The Medical Laboratory Workflow. While patients and clinicians often see only the final test report, each laboratory results is the product of a complex, tightly connected sequence of processes that begin long before a sample reaches an analyser and continue well after a result is released.
Modern medical laboratories are no longer simple testing facilities. They operate as high reliability, safety critical systems, generating the majority of objective data used in clinical decision making. In this environment, workflow failures are not merely operational inefficiencies, they can translate directly into delayed diagnoses, inappropriate treatment, increased healthcare costs and patient harm. Understanding how laboratory workflows function, where they are vulnerable and how they are governed is therefore essential for clinicians, lab professionals and healthcare leaders.
Traditionally, the medical laboratory workflow is described across these interconnected phases: pre- analytical, analytical and post analytical. Although this structure appears linear on paper, real world laboratory operations are dynamic and interdependent.
Pre-analytical phase
The pre-analytical phase is the first and most error prone stage of the medical laboratory workflow. It encompasses every step from clinicians decision to request a test through to arrival, triaging and preparation of sample in the laboratory. Although many of these processes occur outside the laboratory’s physical environment, their quality directly determines the reliability of all subsequent analytical and post analytical results.
From a leadership and patient safety perspective, the pre analytical phase represents a critical zone of shared responsibility, where failures often arise at handover points between clinical, collectors, couriers and lab staff.
1. Requesting tests
The diagnostic process starts with the medical practitioners ordering required tests based on patients history, clinical symptoms presented by the patient and differential diagnoses. Inappropriate testing whether excessive, insufficient or incorrect creates downstream inefficiency, interpretive noise and patient risk.
2. Sample collection
The samples are either collected by the requesting practitioners in clinics/hospitals or by a phlebotomist located at the collection centres. Different samples are collected based on the tests requested. Eg: blood is collected in various tubes (SST, plain, EDTA, K2 EDTA or citrate tubes) according to the order of draw.
Correct phlebotomy technique and appropriate tube selection are critical to maintain sample integrity. Errors at this stage can result in hemolysis, clotting, contamination, insufficient volumes which often necessitates recollection and delaying patient care.
3. Sample transportation
Following collection, specimens are transported from clinics and collection centres to the medical laboratory by dedicated courier services. During transit the sample must be maintained under controlled environmental conditions to preserve analyte stability.
Factors such as temperature fluctuations, excessive agitation, prolonged transit times, or exposure to light can compromise sample quality even before analytical testing begins. Although transportation can be outsourced, labs retain responsibility for defining transport requirements, monitoring performance and ensuring that specimens meet acceptance criteria on arrival. Increasingly, transport processes are being recognised as an extension of the lab workflow rather than an external service.
4. Sample registration
The samples are delivered to the specimen reception where they are sorted into different categories based on the types of samples and the tests requested. Each specimen is checked for identification errors, suitability and compliance with acceptance criteria before being registered into the laboratory system.
During registration, samples are assigned unique identifiers, barcoded and electronically tracked before being routed to the appropriate laboratory departments. Errors at this stage such as mislabelling or incorrect data entry can have serious downstream consequences, reinforcing the importance of robust verification and tracking systems.
5. Sample preparation
Once registered, samples undergo preparation according to laboratory specific standard operating procedures. This may include centrifuge of serum separator or citrate tubes, aliquoting of serum or plasma form specialised testing, or direct analysis of samples tested as received.
From a workflow perspective, sample preparation represents the final opportunity to identify pre analytical issues before testing begins.
Future insight of Pre-analytical phase
The pre-analytical phase is undergoing a rapid transformation driven by advances in automation, digitalisation and artificial intelligence, shifting it from a traditionally manual and error prone process into an increasingly intelligent and proactive system. Innovations such as patient led self sampling and automated blood collection technologies are expanding access to diagnostics while reducing dependence on conventional collection workflows. AI assisted phlebotomy devices, including autonomous ultrasound guided systems are improving first attempt success rates and minimising patient discomfort and blood loss, particularly in high risk and difficult access populations.
Digitalisation of pre-analytical processes is further enhancing safety and traceability. Machine learning-enhanced optical character recognition is being applied to digitise handwritten test requests, reducing transcription errors, while AI-driven image recognition systems can detect improper labelling, haemolysis, clot formation, and insufficient sample volume in real time often before samples proceed to analysis. During transportation, intelligent monitoring systems are emerging that continuously track environmental conditions such as temperature and transit time, enabling early identification of samples at risk of degradation.
At a systems level, demand management strategies are evolving beyond rule-based algorithms toward machine learning and natural language processing models trained on patient demographics, clinical information, and historical testing patterns. These tools show strong potential in reducing both over- and under-utilisation of laboratory tests, supporting more clinically appropriate requesting. In parallel, advanced machine learning models including neural networks, extreme gradient boosting, and deep learning techniques have demonstrated high accuracy in detecting specimen mix-ups, wrong-blood-in-tube events, and clotting abnormalities using routine haematology and chemistry data.
Together, these emerging technologies signal a shift toward intelligent pre-analytical automation, where AI-enabled platforms integrate specimen verification, condition assessment, and dynamic routing with minimal human intervention. Rather than replacing laboratory professionals, these systems augment human decision-making, enabling earlier error detection, reduced diagnostic risk, and more resilient pre-analytical workflows. For laboratory leaders, the challenge lies not in adopting individual technologies, but in governing their safe, ethical, and clinically meaningful integration into everyday diagnostic practice.
Analytical Phase
The analytical phase represents the core technical engine of the medical laboratory workflow, where biological specimens are transformed into quantitative and qualitative results that directly inform clinical decision making. This phase encompasses a highly controlled environment involving advanced instrumentation, automated systems, reagent and consumable management, quality control processes and ongoing calibration and maintenance activities. While often perceived as the most technically stable stage of laboratories testing, the analytical phase is also where labs must continuously balance throughput demands with analytical accuracy, precision and reliability.
Modern laboratories operate across multiple specialised departments including haematology, clinical chemistry, immunology, transfusion science, microbiology, molecular pathology, histopathology and cytogenetics. Each with distinct analytical workflows, risk profiles and quality requirements. Despite these differences, all analytical processes share the common objective: generation of results that an are scientifically valid, clinically meaningful and safe for patient care. Achieving this objective depends not only on technology but on robust quality systems, skilled professional over sight and leadership driven governance frameworks that ensure analytical performance is maintained under increasing workload, automation and complexity.
6. Instrumentation and Automation
This forms the backbone of the analytical phase, enabling labs to meet growing testing demands while maintaining accuracy, consistency and efficiency. Modern medical laboratories deploy a diverse range of analytical platforms, selected not only based on test type but also on testing volume, turnaround time requirements, clinical urgency and risk profile. As diagnostic demand continue to rise, automation plays a critical role in scaling laboratory operations while reducing manual handling and variability.
7. Maintenance of the Analysers
To ensure reliable analytical performance, laboratory analysers undergo routine maintenance in accordance with manufacturer recommendations and laboratory defined procedures. This typically includes daily start up checks, alongside scheduled weekly or monthly maintenance activities. Preventative maintenance, corrective servicing and structured troubleshooting are essential to ensure instruments continue to operate within specified result integrity.
Maintenance activities are not merely technical tasks, they are integral components of quality management systems. Failure to adhere to maintenance schedules can result in undetected analytical drift, increased downtime and compromised result integrity.
8. Running QC
Before patient samples are analysed, Quality Controls (QC) are run to check if the machines are producing accurate and precise results. The frequency of IQC testing varies depending on analyser usage, test stability and lab policy, but commonly run at the start of the day and every 4-6 hours as well as before the introduction of new test batches and specialised assays.
When QC results fall outside defined acceptance limits, controls are repeated to differentiate between random and systematic errors. Trend analysis is performed to identify shifts or gradual changes in analytical performances using Westgard rules and Levy-jenning charts using a specialised QC software or on the instruments. reagent integrity, expiry dates and lot changes are reviewed and recalibration is undertaken when new reagent lots are introduced or when troubleshooting is required. Patient testing is paused until the underlying is identified and resolved.
9. External quality assessment
In addition to internal quality control, laboratories participate in external quality assessment programs. These inter-laboratory comparison schemes allow laboratories to benchmark analytical performance against peers, identify systematic bias, and demonstrate ongoing competence. Participation in EQA programs is a core requirement of accreditation standards and provides an external validations of analytical accuracy and comparability.
10. Sample analysis
After the samples, reagents and quality control are loaded onto the analyser, the tests required to be run on the sample are communicated to the analyser via a middleware that connects it with the Laboratory information system . The analyser performs the required tests and transmits the results to the middleware and this is where the tests are troubleshooted if any alarm is generated.
Despite the increased automation, the analytical phase remains highly dependent on professional judgement at critical decision points. Laboratory scientists are required to interpret QC trends, assess instrument behaviour and determine when to repeat testing, recalibrate systems or escalate issues. When quality control results are out of range or instrument exhibits atypical behaviour, human oversight becomes essential to prevent the release of unreliable results.
Advanced and Innovative approaches: Analytical phase
The analytical phase is rapidly evolving through the integration of robotics, artificial intelligence and predictive analytics, transforming traditional laboratory testing into a more automated, intelligent and anticipatory system. Robotic platforms, increasingly embedded across high throughput disciplines like clinical chemistry, immunology and haematology are being used for automated sample handling, reagent loading, aliquoting and analyser interfacing.
Beyond physical automation, predictive analytics is emerging as critical enabler of advanced quality assurance and clinical insight. Machine learning models are being developed to analyse internal quality control data, analyser performance metrics and historical testing patterns to identify early signs of instrument drift, reagent instability or process deviation often before conventional quality control rules are breached. Moving from reactive error detection to proactive risk identification strengthening analytical reliability.
At clinical level, predictive models applied to analytical data are also supporting early disease detection and risk stratification. In areas such as predicting the probability of sepsis or cardio vascular disease, machine learning models trained on laboratory results along with clinical symptoms, and other data demonstrating the potential to predict these conditions more accurately than single parameter assessment. In molecular genetics, advanced analytical models integrating lab findings with genomics and clinical datasets are enabling more precise disease classification and personalised diagnostic insights.
Computer Vision (CV) and Natural Language Processing (NLP) are rapidly extending the analytical capabilities of medical labs. In haematology computer vision based algorithms are being applied to digital blood films to assist with cell classification, flag abnormal morphologies and detect parasites such as Plasmodium species with high sensitivity. In parallel, digital pathology is transforming anatomical pathology and cytology workflows. AI driven image analysis systems are increasingly used to detect and characterise malignancies by analysing the whole side images at scale.
NLP technologies further enhance the analytical workflow by addressing a long standing source of risk: unstructured and handwritten requests. Machine learning enhanced optical character recognition and NLP models can accurately digitalise handwritten forms, extract clinical context, reducing transcription errors and ensuring that no investigations are missed.
Post Analytical
The post analytical phase represents the final and clinically decisive stage of the medical laboratory workflow. It begins once analytical phase is complete and extends through result validation, reporting, communication and clinical interpretation. While no further testing occurs here the post analytical phase is where the value of laboratory medicine is ultimately realised.
11. Result validation
Following analysis, patient results are complied by the lab information systems are made available to the scientists, pathologists and authorised clinicians. In many modern labs, results that fall within the predefined reference ranges and that fall within predefined reference ranges and are generated without analyser flags or quality concerns may be released automatically through validated auto-verification rules. This enables timely reporting while maintaining efficiency in high throughput environments.
When results are abnormal, inconsistent or associated with analyser flags, they require professional review and validation. Laboratory scientists assess these results in the clinical context of quality control performance, delta checks, patient history and analytical limitations. Where appropriate, pathologist input may be sought, particularly for complex, critical or diagnostically significant findings. This step ensures that results released to clinicians are not only analytically correct but clinically credible.
12. Data, Specimen and Waste Stewardship
Beyond result release, the post analytical phase encompasses the responsible stewardship of laboratory data, clinical material and biological waste.
Laboratory examination results form part of the patient’s permanent health record and must be retained in accordance with regulatory, accreditation and medico-legal requirements. Secure storage, controlled access and data integrity are essential to support continuity of care, clinical audits, quality improvement initiatives and legal accountability.
Laboratories are also responsible for the retention and appropriate storage of clinical specimens and materials for defined periods, depending on discipline and regulatory guidance. This may include blood samples, tissue blocks, histology slides, micro isolates and molecular extracts. Proper storage conditions such as temperature control, traceability, and environmental monitoring ensure that samples remain suitable for repeat testing, confirmatory analysis or investigative review when required.
Laboratories must manage the safe disposal of biological, chemical and sharps waste generated during testing. Waste disposal practices are governed by occupational health and safety standards, infection control policies and environmental regulations. Effective waste management protects laboratory staff and the wider community while also supporting organisational sustainability and risk reduction.
13. Communication and clinical integration
Once validated, laboratory results are communicated to the requesting clinicans, often through integrated electronic health record systems. For critical or life threatening results, additional escalation procedures such as direct verbal notification are followed to ensure timely clinical action. Effective communication at this stage is essential, as delays or misunderstandings can have direct implications for patient outcomes.
Clinicians then integrate laboratory findings with clinical assessment, imaging and patient history to establish diagnosis, access disease severity and guide treatment decisions. In this way, the post analytical phase serves as the interface between laboratory science and patient care, translating numerical data into meaningful clinical insights.
From a leadership and governance standpoint, the post analytical phase extends beyond result release. It includes monitoring turnaround times, managing critical result reporting, addressing clinical queries and processing test adds, reviewing post analytical quality indicators such as amended reports or communication errors. Increasingly, laboratories are also exploring decision support tools and structured interpretive comments to enhance results clarity and clinical relevance.
Intelligent Decision Support in the Post analytical phase
The post analytical phase is increasingly supported by decision algorithm models, AI assisted interpretive commenting and computerised clinical decision support systems (CDSS) that enhance how laboratory results are aggregated, interpreted and reported. Decision algorithms apply rule based and probabilistic logic to laboratory data such as reference ranges, delta checks, clinical thresholds and clinical context to standardise reporting and prioritise clinically significant findings.
AI assisted commenting builds on this foundation by generating context aware interpretive notes that synthesise multiple analyses, prior results and relevant clinical information. Rather than static or generic comments, these systems can tailor guidance based on disease probability, risk stratification and test limitations, improving clarity for clinicians and reducing interpretive variability. CDSS platforms further extend this capability by integrating lab data with EHR, imaging, genomic information to deliver real time alerts, recommendations and follow up testing pathways at the point of care.
Interactive machine learning tools play a complementary role by improving data aggregation and insight generation. Dashboards and visual analytics allow laboratory professionals and clinicians to explore trends, compare cohorts and interrogate results dynamically, supporting audits, quality improvements and clinical decision making.
Together these technologies transform labs into an active central intelligence system, where data driven insights, human expertise and governance frameworks converge to improve diagnostic accuracy, efficiency and patient outcomes.
References:
Alsaedi, A. S. A., Alharbi, N. A. H., Alamri, T. D., Alawamer, A. M., Al Jabri, A. T., Homeda, A., Alsarrani, F. A. H., & Alsharef, A. F. (2024). The Role of Predictive Analytics in Theoretical Modeling of Clinical Laboratory Workflow Optimization. Journal of International Crisis and Risk Communication Research,7(S6), 1864.
Plebani, M. (2024). Harmonizing the post-analytical phase: focus on the laboratory report. Clinical Chemistry and Laboratory Medicine (CCLM), 62(6), 1053-1062.
Plebani, M., Scott, S., Simundic, A.-M., Cornes, M., Padoan, A., Cadamuro, J., Vermeersch, P., Çubukçu, H. C., González, Á., & Nybo, M. (2025). New insights in preanalytical quality. Clinical Chemistry and Laboratory Medicine (CCLM), 63(9), 1682-1692.

Great work
Excellent work ❤
Thank you!
It’s actually a cool and useful piece of info.
I’m satisfied that you simply shared this helpful information with us.
Please stay us informed like this. Thanks for sharing.
You’re so awesome! I do not think I have read through a single thing like
this before. So great to find another person with genuine thoughts on this subject.
Really.. many thanks for starting this up. This site is one thing that is needed on the web, someone with a little originality!
Love the way you have presented this information, very easy to understand and follow. Thank you!