In medical laboratories, the processing of patient samples occurs across three distinct phases: Pre-analytical, Analytical and Post- analytical phases.
This articles focuses exclusively on the traditional pre-analytical workflow as it operates in most medical laboratories today. The pre-analytical phase encompasses all activities from test ordering and specimen collection through to sample handling, transportation and initial processing prior to analysis.
This phase begins when a diagnostic test is ordered by the doctor and the sample is collected by nurses in the hospital or phlebotomists in the collection centres. These samples are delivered to the medical laboratories via the laboratories private transport system or via pneumatic tube systems commonly found in hospital laboratories. Upon arrival, all specimens are received and processed through the laboratory’s sample reception area, marking the formal entry of the sample into the laboratory testing pathway.
Triage:
The first step following sample receipt is triage, which involves sorting specimens according to urgency, sample type, and required processing pathways. In most laboratories, samples are prioritised based on clinical urgency. This includes specimens associated with urgent test requests, such as cardiac biomarkers (e.g., troponin for suspected acute coronary events), samples from patients scheduled for surgery or currently in the operating theatre, and specimens with limited stability that require timely processing, such as coagulation samples, which typically need to be analysed within 2–4 hours of collection.
In addition to urgency-based prioritisation, samples are segregated according to specimen type. Routine samples include blood, swabs, urine, and faecal specimens, while cytology samples commonly include cervical smears and urine specimens, and histology samples consist of skin and other tissue biopsies. Grouping and pooling similar sample types during triage facilitates efficient registration and downstream processing within the laboratory.
Pre-Sorting
Medical laboratories may receive pre-barcoded specimens from external clinics and collection centres. During the pre-sorting stage, these samples are audited and cross-checked to confirm that the specimens received correspond to the tests requested. This includes verifying sample type, labelling, and completeness of accompanying documentation.
Request forms are then scanned to initiate registration within the laboratory information system, after which samples are directed to their respective laboratory departments for further pre-analytical processing.
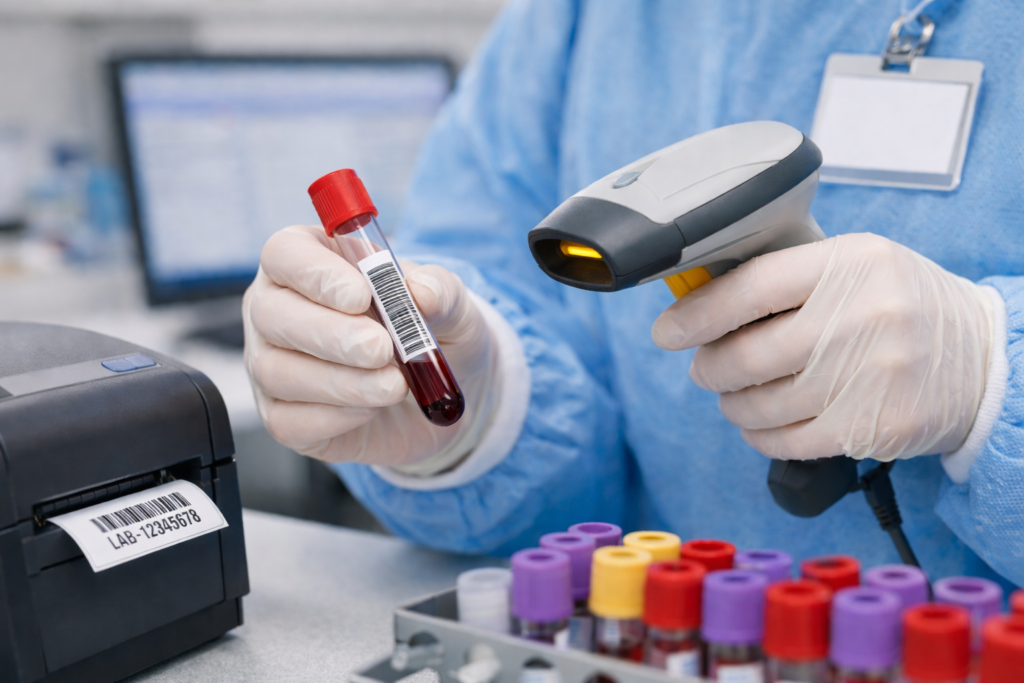
Barcoding
Routine blood, cytology, histology, and 24-hour urine specimens are barcoded in accordance with laboratory-specific procedures to ensure accurate identification and traceability. Laboratories may utilise either pre-printed barcode labels or computer-generated barcodes produced at the time of sample receipt.
Laboratory test guides are consulted during this stage to confirm any special requirements associated with the requested tests. Relevant test information and sample details are entered into the request documentation and scanned to enable registration within the laboratory information system. Once barcoding and registration are complete, samples are routed to their respective laboratory departments for further processing.
Special requirement
Samples that do not meet pre-analytical acceptance criteria, such as incorrect sample type, labelling errors, missing specimens, absent request forms, or inappropriate transportation conditions may require recollection in accordance with laboratory protocols.
Certain tests have specific pre-processing requirements. For example, assays such as lupus anticoagulant testing may require double centrifugation and aliquoting, while some molecular investigations, including viral load testing, require the collection of separate specimens. Samples designated for referral or shared testing are typically aliquoted and dispatched to external laboratories following established procedures, including the use of sterile techniques to maintain sample integrity.
Many biochemical investigations require centrifugation to separate blood components prior to analysis. Tests such as electrolytes, liver function tests, and renal function tests are commonly performed on serum, whereas coagulation assays require plasma samples.
Registration
Following barcoding and scanning of request forms, the requested tests are registered into the laboratory information system. This step involves verifying that patient demographic details are accurately recorded to ensure correct patient identification and traceability throughout the testing process.
The requested investigations are then entered into the system using the appropriate test codes, along with the details of the requesting clinician or healthcare provider and the billing details. Accurate entry of requester information is essential to ensure correct reporting and timely transmission of results to the appropriate clinical teams.
Dispatch
Most medical laboratories perform commonly requested investigations on site, while specialised tests are referred to hospital-based or external referral laboratories. Following registration, samples designated for referral testing are packaged in accordance with transport and temperature requirements, typically placed in appropriate containers within chilled transport systems. These samples are scanned into dedicated send-away or referral tracking systems to ensure traceability throughout transit and testing
The pre-analytical phase plays a critical role in the overall laboratory testing process, as it encompasses key steps such as sample identification, registration, and handling prior to analysis. Errors occurring during this phase can compromise the accuracy and reliability of laboratory results. Given that laboratory findings directly inform clinical diagnosis, treatment decisions, and patient management, inaccuracies arising from pre-analytical processes may have significant implications for patient care.
Click here to read more about: Medical Laboratory Workflow
In the future articles, we will explore how automation, artificial intelligence, quality systems and leadership frameworks are being applied to the pre analytical phase to reduce errors and improve patient outcomes.
